
Cost-effectiveness analysis of oral antiviral therapy in patients with indeterminate chronic hepatitis B infection
Highlight box
Key findings
• The study found that antiviral therapy (AVT) for patients with indeterminate phase-chronic hepatitis B (IP-CHB) is highly cost-effective, significantly reducing the incidence of cirrhosis, hepatocellular carcinoma, and hepatitis B virus (HBV)-related mortality, while adding 1.4541 quality-adjusted life years (QALYs) per patient at an incremental cost-effectiveness ratio (ICER) of $253.51 per QALY. This highlights the need for updated clinical guidelines to include AVT for IP-CHB patients.
What is known and what is new?
• IP-CHB patients face high risks of liver inflammation, fibrosis, and progression to HCC. While AVT is known to reduce complications in other CHB stages, its cost-effectiveness in IP-CHB was previously unclear. This study confirms that AVT is highly cost-effective, reducing cirrhosis, HCC, and liver-related mortality over 50 years, with an ICER of $253.51 per QALY, far below China’s 2023 GDP per capita threshold ($12,690.1).
• The study demonstrates AVT’s cost-effectiveness for IP-CHB by adding 1.4541 QALYs per patient, reducing 11,581, 15,436, and 15,112 cases of compensated cirrhosis, decompensated cirrhosis, and HCC, respectively, and preventing 28,923 HBV-related deaths in 100,000 patients over 50 years.
What is the implication, and what should change now?
• AVT for IP-CHB patients not only improves survival and quality of life but also alleviates the economic burden of HBV-related diseases. This highlights the need to include IP-CHB patients in AVT eligibility criteria globally.
• Clinical guidelines should be updated to recommend AVT for IP-CHB patients, considering their significant risk of progression and the demonstrated cost-effectiveness of treatment. Healthcare policymakers should prioritize funding and access to affordable AVT.
Introduction
According to the World Health Organization (WHO), there were 296 million chronic hepatitis B virus (HBV) infections worldwide in 2019 (1), with 80.65 million new cases reported annually (2). An estimated 3.31 million deaths can be attributed to HBV-related cirrhosis and chronic liver disease (1,3). China has the most considerable burden of HBV infection globally, with an HBV prevalence of 5.6% in 2022 (4), translating to 79.75 million hepatitis B surface antigen (HBsAg)-positive individuals (4). In China, only 24% (19.13 million) of chronic HBV patients were diagnosed (4), and although 33.87 million were eligible for treatment, only 5.08 million (15%) received antiviral therapy (AVT) (4). The WHO has set a goal of “eliminating viral hepatitis as a public health hazard by 2030”, aiming to reduce new infections by 90% and the case fatality rate by 65% (5). In China, according to the 2016 national cancer statistics, liver cancer ranked fourth in incidence and second in mortality among malignant tumors (6). An estimated 367,700 new cases of liver cancer are reported annually (7).
According to the Guideline for Chronic Hepatitis B (CHB) (2022 edition) (8), the natural history of chronic HBV infection can be divided into four stages: hepatitis B e antigen (HBeAg)-positive chronic HBV infection, HBeAg-positive CHB, HBeAg-negative chronic HBV infection, and HBeAg-negative CHB. However, the condition of CHB patients can be complex, and a significant proportion cannot be classified into these four categories, leading to what is known as the indeterminate phase (IP). Patients in this phase have a history of infection lasting more than six months, but their HBV deoxyribonucleic acid (DNA) and alanine transaminase (ALT) patterns differ from those in the traditional stages of chronic HBV infection (9). This is why the IP is also referred to as the grey zone (10-13). It has been reported that individuals in the IP represent up to 27.8% to 59.5% of patients with chronic HBV infection (9-16). Liver biopsy studies have shown that significant necrotic inflammation is present in 29.2% to 84.4% of patients in the IP, while significant liver fibrosis is observed in 34.0% to 73.3% of these patients (11,12,16-18). Notably, significant necrotic inflammation and/or significant liver fibrosis were previously identified in 49.7% to 91.1% of patients in the IP (11,12,16,17). Nevertheless, there are no established recommendations regarding whether patients in the IP should receive AVT (19).
Oral AVT can reduce the burden of liver cirrhosis by approximately 74% (20). Several studies have indicated that patients in the IP have a higher risk of developing hepatocellular carcinoma (HCC) (9,21-23). However, a study by Huang et al. has indicated that AVT for patients in the IP can reduce the risk of liver cancer by 70% (24). Whether oral AVT is cost-effective for patients in the IP remains unclear. Therefore, in the present study, we conducted a cost-effectiveness analysis (CEA) of AVT for CHB patients in the IP. We present this article in accordance with the CHEERS reporting checklist (available at https://tgh.amegroups.com/article/view/10.21037/tgh-24-163/rc).
Methods
Overview of the cost-effectiveness model
Herein, we conducted a CEA using a Markov model to compare two scenarios for patients with IP-CHB who do not meet the criteria for oral AVT: patients who received oral AVT (scenario I) vs. those who did not receive oral AVT, reflecting the current clinical practice (scenario II) (Figure 1).
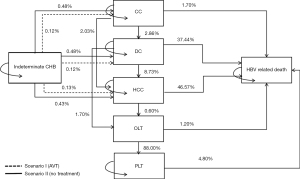
The model consisted of seven health states that patients with IP-CHB may progress through until the end of life. These health states, depicted in Figure 1, represent the entire clinical course of disease progression: (I) IP-CHB; (II) compensated cirrhosis (CC); (III) decompensated cirrhosis (DC); (IV) HCC; (V) orthotopic liver transplantation (OLT); (VI) post-orthotopic liver transplantation (PLT); and (VII) HBV-related death. A hypothetical cohort of patients aged 34 years (n=100,000) was created. Each cycle period was one year, and patients were distributed across each state according to transition probabilities at the end of each cycle. The base case analysis used a 50-year analytic period and was conducted from the healthcare system’s perspective. A discount rate of 5% was applied, following the Chinese guidelines for CEA, along with half-cycle corrections for costs and quality-adjusted life years (QALYs). In 2023, the gross domestic product (GDP) per capita of China was approximately $12,690.1 (25), which served as the threshold for the willingness to pay (WTP).
Effects in this study are expressed in QALYs, and the CEA also assessed the incremental cost-effectiveness ratio (ICER). The Markov model was calculated and implemented using TreeAge Pro 2019 (TreeAge Software, Williamstown, MA, USA) and Microsoft Excel 2019.
Transition probabilities
The transition probabilities for each cycle were crucial in determining the distribution of patients across the seven specified health states. These probabilities were derived from previously published studies involving patient populations comparable to those in the current research. The transition probabilities are illustrated by arrows in Figure 1.
The probability of IP-CHB progressing to liver cirrhosis without oral AVT was 0.96% (9). Since cirrhosis includes both CC and DC, we hypothesized that each type accounted for half of the cases, resulting in a probability of 0.48% for either CC or DC. Oral antiviral treatment can reduce the burden of liver cirrhosis by 74% (20). Therefore, the probability of IP-CHB patients receiving oral AVT developing either CC or DC was only 0.12%. The probability of IP-CHB progressing to HCC without AVT was 0.43%, while the probability for those undergoing oral AVT was 0.13% (9,20,24). However, the study has indicated that AVT for patients in the IP can reduce the risk of liver cancer by 70% (24). The other transition probabilities were obtained from published medical literature and public databases, along with parameterized treatment costs and other model parameters (Table 1).
Table 1
| Parameters | One-way sensitivity analysis | PSA | Data source | ||
|---|---|---|---|---|---|
| Value | Lower limit | Upper limit | |||
| Transition probabilities (%) | |||||
| From indeterminate CHB (scenario II) to | |||||
| CC | 0.48 | 0.38 | 0.58 | Point-estimate | (9) |
| DC | 0.48 | 0.38 | 0.58 | Point-estimate | (9) |
| HCC | 0.43 | 0.34 | 0.52 | Point-estimate | (9) |
| From indeterminate CHB (scenario I) to | Point-estimate | ||||
| CC | 0.12 | 0.10 | 0.14 | Point-estimate | (9,20) |
| DC | 0.12 | 0.10 | 0.14 | Point-estimate | (9,20) |
| HCC | 0.13 | 0.10 | 0.16 | Point-estimate | (9,20,24) |
| From CC to | |||||
| DC | 2.86 | 2.29 | 3.43 | Point-estimate | (26) |
| HCC | 2.03 | 1.62 | 2.44 | Point-estimate | (26) |
| HBV-related death | 1.70 | 1.20 | 4.80 | Beta (2.82, 163.29) | (27-29) |
| From DC to | |||||
| HCC | 8.73 | 6.98 | 10.48 | Point-estimate | (26) |
| OLT | 1.70 | 0.10 | 4.20 | Beta (1.25, 72.03) | (30) |
| HBV-related death | 37.44 | 37.26 | 55.88 | Point-estimate | (26) |
| From HCC to | |||||
| OLT | 0.60 | 0.10 | 3.00 | Beta (1.43, 236.13) | (30) |
| HBV-related death | 46.57 | 37.26 | 55.88 | Point-estimate | (26) |
| From OLT to | |||||
| PLT | 88.00 | 86.80 | 89.20 | Point-estimate | (31) |
| HBV-related death | 1.20 | 10.80 | 13.20 | Point-estimate | (31) |
| From PLT to | |||||
| HBV-related death | 4.80 | 4.30 | 5.30 | Point-estimate | (32) |
| Utility weight | |||||
| Indeterminate CHB | 0.79 | 0.74 | 0.84 | Beta (51.63, 13.73) | (33-40) |
| CC | 0.73 | 0.69 | 0.78 | Beta (89.19, 32.99) | (33-40) |
| DC | 0.65 | 0.63 | 0.67 | Beta (369.04, 198.71) | (33-40) |
| HCC | 0.38 | 0.36 | 0.41 | Beta (223.44, 364.56) | (33-40) |
| OLT | 0.67 | 0.64 | 0.69 | Beta (163.93, 80.74) | (33-40) |
| PLT | 0.55 | 0.49 | 0.61 | Point-estimate | (33,41) |
| Costs ($USD) | |||||
| CC | 4,330 | 3,460 | 5,200 | Gamma (24.77, 0.0057) | (42) |
| DC | 5,523 | 4,420 | 6,630 | Gamma (25.21, 0.0046) | (42) |
| HCC | 7,301 | 5,840 | 8,760 | Gamma (23.69, 0.0032) | (42) |
| OLT | 35,701 | 28,560 | 42,840 | Gamma (27.11, 1.08) | (42) |
| PLT | 8,393 | 6,710 | 10,070 | Gamma (24.37, 0.0029) | (43) |
| Indeterminate CHB AVT treatment | 236.01 | 215.08 | 256.93 | Point-estimate | |
| Discount rate: costs (%) | 5 | 0 | 5 | (44) | |
| Discount rate: health outcomes (%) | 5 | 0 | 5 | (44) | |
AVT, antiviral therapy; CC, compensated cirrhosis; CHB, chronic hepatitis B; DC, decompensated cirrhosis; HBV, hepatitis B virus; HCC, hepatocellular carcinoma; OLT, orthotopic liver transplantation; PLT, post-orthotopic liver transplantation; PSA, probabilistic sensitivity analyses.
Cost and utility estimates
From the perspective of the Chinese health system, this study estimates only the direct medical costs, which include the cost of oral antiviral drugs, the direct costs of biannual tests and examinations, and the direct medical costs associated with hepatitis B-related diseases. Three antiviral therapies, namely entecavir (ETV) (0.5 mg tablet taken once daily), tenofovir disoproxil fumarate (TDF) (300 mg tablet taken once daily), and tenofovir alafenamide fumarate (TAF) (25 mg tablet taken once daily), are recommended by the Guideline for Chronic Hepatitis B (2022 edition) (8). Currently, generic versions of ETV, TDF, and TAF are much cheaper and widely prescribed. The median cost of these generic versions is 202.39 CNY per year. The cost of AVT-related serum measurements and examinations, such as liver biochemistry, HBV DNA, HBV markers, α-fetoprotein, and ultrasound, amounts to 1,394.40 CNY per year. Therefore, the total cost related to AVT for patients in the IP is estimated to be around 202.39+1,394.40=1,596.79 CNY per year. We used the average exchange rate of 6.7659 RMB to 1 USD ($), according to the rates offered by the Bank of China in 2023, which amounted to 236.01 USD.
Next, we hypothesized that the utility of indeterminate CHB was 0.79, which is equivalent to that of CHB. The other costs and utilities were obtained from recent CEA of Chinese individuals with CHB (45) and were used to estimate health state utility (Table 1).
Statistical analysis
Statistical analyses were conducted using TreeAge Pro 2019 and Microsoft Excel 2019, including probabilistic modeling of transition probabilities (Table 1), one-way deterministic sensitivity analysis (parameter ranges derived from literature extrema), and probabilistic sensitivity analysis (PSA) (1,000 Monte Carlo simulations). All parameters were estimated via literature synthesis or expert consensus, following China’s Pharmacoeconomic Guidelines (44) for discounting (5%) and half-cycle correction.
Sensitivity analysis
To avoid uncertainty in the analytical results and mitigate the risk of potential bias, this study employed one-way deterministic sensitivity analyses and PSA to assess the robustness of the model. The minimum and maximum values were determined by obtaining the lower and upper limits in the one-way deterministic sensitivity analyses. The findings were presented using a tornado diagram. PSA characterized the combined uncertainty of all model parameters based on 1,000 Monte Carlo simulations, generating a cost-effectiveness acceptability curve and an incremental cost-effectiveness scatter plot. These indicated the probability that each alternative would be the most cost-effective treatment as a dominant strategy at various WTP thresholds. Herein, we set the WTP threshold at one time the GDP per capita.
Results
Base-case analysis
From the healthcare system’s perspective, oral AVT for patients with IP-CHB provided greater health benefits at a higher cost than no treatment (see Table 2). Over a 50-year period, the estimated total cost per patient was $5,777.57 for the AVT group (scenario I) and $5,408.95 for the no-treatment group (scenario II), resulting in a cost difference of $368.6225. The simulation indicated that patients receiving oral AVT gained an additional 1.4541 QALYs (14.1520 vs. 12.6979) compared to those who did not receive treatment. As a result, the ICER for AVT was calculated to be $253.51 per QALY.
Table 2
| Parameter | Scenario I (AVT) | Scenario II (no treatment) | Incremental value |
|---|---|---|---|
| NMB† | 556,652.13 | 499,233.39 | 57,418.74 |
| ICER‡ | 253.51 | ||
| Cost (discounted)† | 5,777.57 | 5,408.95 | 368.6225 |
| Effectiveness (discounted)§ | 14.1520 | 12.6979 | 1.4541 |
| Incidence of CC among 100,000 patients with IP-CHB | 5,609 | 17,190 | −11,581 |
| Incidence of DC among 100,000 patients with IP-CHB | 7,354 | 22,790 | −15,436 |
| Incidence of HCC among 100,000 patients with IP-CHB | 8,358 | 23,470 | −15,112 |
| Incidence of OLT among 100,000 patients with IP-CHB | 352 | 1,070 | −718 |
| Number of HBV-related deaths among 100,000 patients with IP-CHB | 14,652 | 43,575 | −28,923 |
†, The unit for NMB and cost (discounted) is USD. ‡, The unit for ICER is USD/QALY. §, The unit for effectiveness (discounted) is QALY.ICER = Incremental cost (discounted)/Incremental effectiveness (discounted). AVT, antiviral therapy; CC, compensated cirrhosis; DC, decompensated cirrhosis; HBV, hepatitis B virus; HCC, hepatocellular carcinoma; ICER, incremental cost-effectiveness ratio; IP-CHB, indeterminate phase-chronic hepatitis B; NMB, net monetary benefit; OLT, orthotopic liver transplantation; QALY, quality-adjusted life year.
In a cohort of 100,000 patients with IP-CHB receiving oral AVT, a significant reduction in the incidence of cirrhosis-related complications and liver disease-related mortality could be achieved. Over a 50-year period of continued AVT, it is estimated that 11,581 cases of CC, 15,436 cases of DC, and 718 cases of OLT could be prevented. These findings underscore the long-term benefits of sustained antiviral treatment in alleviating the burden of advanced liver disease among patients with IP-CHB.
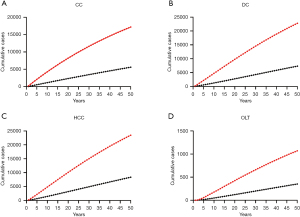
In a cohort of 100,000 patients with IP-CHB, the incidence of HCC over a 50-year period was significantly lower in those receiving oral AVT compared to those who did not receive treatment. In the no-treatment group, there were 23,470 new cases of HCC, whereas only 8,358 cases were observed in the AVT group. This indicates that oral AVT could prevent approximately 15,112 new cases of HCC over 50 years. Furthermore, oral AVT was associated with a substantial reduction in mortality, potentially saving over 30% of patients’ lives. Specifically, the AVT group averted 28,923 deaths, with 14,652 deaths recorded compared to 43,575 deaths in the no-treatment group. These findings underscore the critical role of AVT in reducing both the incidence of HCC and liver disease-related mortality in patients with IP-CHB. In addition, the cumulative cases of new HBV-related diseases would be significantly reduced year by year following the initiation of antiviral treatment (Figure 2). As shown in Figure 2, AVT (black dotted line) significantly reduced the cumulative cases of CC, DC, HCC, and OLT compared to no treatment (red dotted line) over the 50-year period (Figure 2).
One-way sensitivity analysis
The results of the one-way sensitivity analysis, illustrated by the incremental net monetary benefit (INMB) tornado plot, revealed that the most influential variable affecting the INMB was the probability of IP-CHB progressing to DC. As this probability increased from 0.38% to 0.58%, the INMB ranged from $15,956.13 to $20,163.15. The other two significant factors influencing the economic outcomes were the probability of IP-CHB progressing to liver cancer and the utility value associated with IP-CHB. Notably, across all variations in parameter values within their respective upper and lower limits, the economic efficiency of AVT for IP-CHB remained superior to no treatment, with an INMB consistently greater than zero (INMB >0) (Figure 3).

PSA
The economic evaluation of AVT for patients with IP-CHB yielded highly favorable results. Compared to no treatment, AVT was deemed cost-effective with a 100% probability at a WTP threshold of one time the GDP per capita. This threshold, set at $12,690.1 (China’s per capita GDP in 2023), showed that AVT emerged as the preferred option in all simulations for patients with IP-CHB.
As shown in Figure 4A, the incremental cost-effectiveness scatter plot illustrates that 100% of the incremental cost-effectiveness points are positioned below the line, representing one time the GDP per capita, indicating that antiviral treatment is consistently more cost-effective than no treatment. Moreover, the cost-effectiveness acceptability curve reveals that AVT becomes more cost-effective at WTP values above $340 (Figure 4B). These findings confirm that antiviral treatment strategies for patients with IP-CHB provide a significant economic advantage compared to no treatment across various WTP thresholds.

Discussion
Herein, we used a Markov model to perform a CEA of AVT in patients with IP-CHB. This approach allowed for a systematic evaluation of the economic impact and health outcomes of antiviral treatment, offering valuable insights into its efficacy and potential benefits for this patient population. Our findings showed that AVT significantly increases QALYs gained, with the ICER well below one time the GDP per capita. The ICER was calculated at 253.51 USD per QALY, substantially lower than the GDP per capita ($12,690.1), indicating apparent cost-effectiveness. The simulation involving 100,000 IP-CHB cases showed that AVT significantly reduces the incidence of end-stage liver disease and hepatitis B-related mortality compared to no treatment. Our findings suggest that AVT could potentially prevent 27,017 cases of liver cirrhosis and 15,112 cases of liver cancer, thereby easing the economic burden associated with these conditions. Both the one-way and PSA confirmed the robustness of the model’s results. These findings suggest that AVT for patients with indeterminate HBV infection provides substantial cost-effectiveness.
Previous studies have underscored the importance of antiviral treatment for patients with IP-CHB. A significant proportion of these patients, regardless of age, show considerable liver tissue damage, ranging from 55.6% to 64.3% (13,14,17,46). Additionally, the risk of developing HCC in this population is 14 times higher than in patients with inactive HBV infection (1). AVT for individuals with indeterminate HBV infection has been shown to significantly reduce the incidence of both liver cirrhosis (20) and HCC (22,47).
The current analysis demonstrates significant health and economic benefits, offering strong justification for timely antiviral treatment in patients with IP-CHB. Our findings show that treating 100,000 patients over a 50-year period could prevent 42,000 new cases of HBV-related complications and avoid 29,000 HBV-related deaths. The low cost of domestically produced generic antiviral drugs, at just $236.01 USD per year for both medication and monitoring, further enhances the cost-effectiveness of treatment. Furthermore, the ICER in this study is notably lower than that for treating all HBsAg-positive patients (48), allowing for more targeted and efficient use of AVT. This approach avoids unnecessary resource expenditure and reduces the potential risks of adverse drug reactions, reinforcing that the antiviral treatment strategy evaluated in this study is indeed cost-effective. Jeng et al. (49) proposed that regular monitoring without AVT might be sufficient to prevent adverse outcomes in patients with IP-CHB. This recommendation likely stems from the relatively low probability of HCC development in this population (0.08%) (50) and the fact that progression to HCC generally requires transition to the immune-active phase of CHB. However, the median follow-up period in these studies was only 4.7 years, which may not have been long enough to fully capture the risk of HCC development (31). However, the median follow-up period in the study by Lo et al. was only 4.7 years (31), which may not have been long enough to fully capture the risk of HCC development, as demonstrated in studies with longer observation periods (22). In contrast, another study with a median follow-up of 10 years for patients with indeterminate CHB estimated an HCC progression rate of 0.43%, providing a more reliable assessment of the long-term risk (9). Besides, two studies from Korea (47,51) analyzed disease progression and cost-effectiveness of AVT in patients who did not meet treatment criteria with HBV DNA levels above 2,000 IU/mL (32), including some IP-CHB patients. Our study, however, specifically focused on a cohort entirely comprised of indeterminate CHB patients. Based on these findings, antiviral treatment for indeterminate CHB is strongly recommended and cost-effective.
Nonetheless, this study has several limitations. First, our analysis was conducted from the perspective of the Chinese healthcare system, utilizing cost and effectiveness data specific to China. Healthcare infrastructure, drug pricing, and willingness-to-pay thresholds vary significantly across different countries, which may affect the generalizability of our findings. Future studies could incorporate international comparisons to assess the cost-effectiveness of AVT in diverse healthcare settings. Second, our model may have underestimated the economic burden of the disease, as it did not include the costs associated with originally developed antiviral drugs, travel expenses for appointments, direct non-medical costs, or indirect costs. Moreover, if productivity losses were factored into the calculation of disability-adjusted life years, the cost-effectiveness would likely improve significantly. Furthermore, the analysis did not account for the impact of natural mortality from non-hepatitis B-related diseases. Lastly, the economic burden associated with adverse drug reactions was not considered in this study.
The heterogeneity of IP-CHB underscores the importance of subgroup stratification, particularly between patients with high viral load and normal ALT levels (GZ-C category) and those with low viral load and elevated ALT levels (GZ-D category), as defined by existing classifications (10). However, critical gaps in subgroup-specific data hinder such analyses. For example, while 70% of the indeterminate-phase population may fall into the GZ-C subgroup (9), longitudinal progression data for GZ-D and other subgroups remain limited. This is compounded by a lack of robust transition probabilities (e.g., progression to cirrhosis or HCC) specific to each subgroup, which are essential for stratified cost-effectiveness modeling. Future studies should prioritize real-world data collection on disease trajectories, healthcare utilization, and clinical outcomes across IP-CHB subgroups. Such data would enable the development of dynamic models that account for subgroup-specific risks and treatment responses, thereby refining cost-effectiveness estimates and guiding personalized management.
Conclusions
In conclusion, our study shows that oral AVT for patients with IP-CHB reduces HBV-related disease progression and mortality, alleviating the global burden of HBV in a cost-effective manner.
Acknowledgments
None.
Footnote
Reporting Checklist: The authors have completed the CHEERS reporting checklist. Available at https://tgh.amegroups.com/article/view/10.21037/tgh-24-163/rc
Data Sharing Statement: Available at https://tgh.amegroups.com/article/view/10.21037/tgh-24-163/dss
Peer Review File: Available at https://tgh.amegroups.com/article/view/10.21037/tgh-24-163/prf
Funding: The study was funded by
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at https://tgh.amegroups.com/article/view/10.21037/tgh-24-163/coif). M.Z.H. reports the funding from Integrated Chinese and Western Medicine “Flagship” Department (Construction Project) of National Health Commission of the People’s Republic of China and National Administration of Traditional Chinese Medicine. J.S.P. reports the funding from the Natural Science Foundation of China (No. 82372318), the Natural Science Foundation of Fujian Province of China (No. 2022J02030), and the Major Research Project for Young and Middle-aged People of the Health Commission of Fujian Province (No. 2022ZQNZD004). The other authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Hsu YC, Huang DQ, Nguyen MH. Global burden of hepatitis B virus: current status, missed opportunities and a call for action. Nat Rev Gastroenterol Hepatol 2023;20:524-37. [Crossref] [PubMed]
- Cao G, Jing W, Liu J, et al. Countdown on hepatitis B elimination by 2030: the global burden of liver disease related to hepatitis B and association with socioeconomic status. Hepatol Int 2022;16:1282-96. [Crossref] [PubMed]
- Global, regional, and national burden of hepatitis B, 1990-2019: a systematic analysis for the Global Burden of Disease Study 2019. Lancet Gastroenterol Hepatol 2022;7:796-829. [Crossref] [PubMed]
- Global prevalence, cascade of care, and prophylaxis coverage of hepatitis B in 2022: a modelling study. Lancet Gastroenterol Hepatol 2023;8:879-907. [Crossref] [PubMed]
- Organization WH. Global hepatitis report, 2017. Meeting report. [Accessed March 15 2023]. Available online: https://www.who.int/publications/i/item/9789241565455
- Chen W, Zheng R, Baade PD, et al. Cancer statistics in China, 2015. CA Cancer J Clin 2016;66:115-32. [Crossref] [PubMed]
- Zheng RS, Chen R, Han BF, et al. Cancer incidence and mortality in China, 2022. Zhonghua Zhong Liu Za Zhi 2024;46:221-31. [Crossref] [PubMed]
- You H, Wang F, Li T, et al. Guidelines for the Prevention and Treatment of Chronic Hepatitis B (version 2022). J Clin Transl Hepatol 2023;11:1425-42. [Crossref] [PubMed]
- Huang DQ, Li X, Le MH, et al. Natural History and Hepatocellular Carcinoma Risk in Untreated Chronic Hepatitis B Patients With Indeterminate Phase. Clin Gastroenterol Hepatol 2022;20:1803-1812.e5. [Crossref] [PubMed]
- Yao K, Liu J, Wang J, et al. Distribution and clinical characteristics of patients with chronic hepatitis B virus infection in the grey zone. J Viral Hepat 2021;28:1025-33. [Crossref] [PubMed]
- Jiang SW, Lian X, Hu AR, et al. Liver histopathological lesions is severe in patients with normal alanine transaminase and low to moderate hepatitis B virus DNA replication. World J Gastroenterol 2023;29:2479-94. [Crossref] [PubMed]
- Wang J, Yan X, Zhu L, et al. Significant histological disease of patients with chronic hepatitis B virus infection in the grey zone. Aliment Pharmacol Ther 2023;57:464-74. [Crossref] [PubMed]
- Xu X, Wang H, Shan S, et al. The Impact of the Definitions of Clinical Phases on the Profiles of Grey-Zone Patients with Chronic Hepatitis B Virus Infection. Viruses 2023;15:1212. [Crossref] [PubMed]
- Duan M, Chi X, Xiao H, et al. High-normal alanine aminotransferase is an indicator for liver histopathology in HBeAg-negative chronic hepatitis B. Hepatol Int 2021;15:318-27. [Crossref] [PubMed]
- Ju Y, Han G, Zhang P, et al. Staging and clinical characteristics of pregnant women with chronic hepatitis B virus infection: A retrospective cohort study from Nanjing, China. J Obstet Gynaecol Res 2023;49:2427-35. [Crossref] [PubMed]
- Chen S, Dai X, Zhao Y, et al. Clinical Distribution Characteristics and Identification for Significant Liver Inflammation of Patients in Chronic Hepatitis B with Indeterminate Phase. Gastroenterol Res Pract 2023;2023:7264601. [Crossref] [PubMed]
- Liu J, Wang J, Yan X, et al. Presence of Liver Inflammation in Asian Patients With Chronic Hepatitis B With Normal ALT and Detectable HBV DNA in Absence of Liver Fibrosis. Hepatol Commun 2022;6:855-66. [Crossref] [PubMed]
- Lai JC, Wong GL, Tse YK, et al. Histological severity, clinical outcomes and impact of antiviral treatment in indeterminate phase of chronic hepatitis B: A systematic review and meta-analysis. J Hepatol 2025;82:992-1003. [Crossref] [PubMed]
- Guidelines for the Prevention, Diagnosis, Care and Treatment for People with Chronic Hepatitis B Infection (Text Extract): Executive Summary. Infect Dis Immun 2024;4:103-5. [Crossref] [PubMed]
- Marcellin P, Gane E, Buti M, et al. Regression of cirrhosis during treatment with tenofovir disoproxil fumarate for chronic hepatitis B: a 5-year open-label follow-up study. Lancet 2013;381:468-75. [Crossref] [PubMed]
- Choi GH, Kim GA, Choi J, et al. High risk of clinical events in untreated HBeAg-negative chronic hepatitis B patients with high viral load and no significant ALT elevation. Aliment Pharmacol Ther 2019;50:215-26. [Crossref] [PubMed]
- Kim GA, Han S, Choi GH, et al. Moderate levels of serum hepatitis B virus DNA are associated with the highest risk of hepatocellular carcinoma in chronic hepatitis B patients. Aliment Pharmacol Ther 2020;51:1169-79. [Crossref] [PubMed]
- Pak K, Sachar R, Saab S. Projected Mitigation of Hepatocellular Carcinoma in Patients with Chronic Hepatitis B in the Gray Zone and the Immune-Tolerant Phase in the United States. Dig Dis Sci 2025;70:1547-54. [Crossref] [PubMed]
- Huang DQ, Tran A, Yeh ML, et al. Antiviral therapy substantially reduces HCC risk in patients with chronic hepatitis B infection in the indeterminate phase. Hepatology 2023;78:1558-68. [Crossref] [PubMed]
- Statistics NBo. Statistical Bulletin on National Economic and Social Development of the People's Republic of China in 2023. [Accessed February 29 2024]. Available online: https://www.stats.gov.cn/sj/zxfb/202402/t20240228_1947915.html
- Lee H, Jang S, Ahn SH, et al. Cost-effectiveness of antiviral therapy in untreated compensated cirrhosis patient with serum HBV-DNA level < 2000 IU/mL. Hepatol Int 2022;16:294-305. [Crossref] [PubMed]
- Tsai TY, Hung TH, Livneh H, et al. Chinese herbal medicine therapy and the risk of mortality for chronic hepatitis B patients with concurrent liver cirrhosis: a nationwide population-based cohort study. Oncotarget 2018;9:18214-23. [Crossref] [PubMed]
- Chang TT, Gish RG, de Man R, et al. A comparison of entecavir and lamivudine for HBeAg-positive chronic hepatitis B. N Engl J Med 2006;354:1001-10. [Crossref] [PubMed]
- Lai CL, Shouval D, Lok AS, et al. Entecavir versus lamivudine for patients with HBeAg-negative chronic hepatitis B. N Engl J Med 2006;354:1011-20. [Crossref] [PubMed]
- Veldhuijzen IK, Toy M, Hahné SJ, et al. Screening and early treatment of migrants for chronic hepatitis B virus infection is cost-effective. Gastroenterology 2010;138:522-30. [Crossref] [PubMed]
- Lo CM, Fan ST, Liu CL, et al. The role and limitation of living donor liver transplantation for hepatocellular carcinoma. Liver Transpl 2004;10:440-7. [Crossref] [PubMed]
- Wu TJ, Chan KM, Chou HS, et al. Liver transplantation in patients with hepatitis B virus-related hepatocellular carcinoma: the influence of viral characteristics on clinical outcome. Ann Surg Oncol 2013;20:3582-90. [Crossref] [PubMed]
- Toy M, Salomon JA, Jiang H, et al. Population health impact and cost-effectiveness of monitoring inactive chronic hepatitis B and treating eligible patients in Shanghai, China. Hepatology 2014;60:46-55. [Crossref] [PubMed]
- Levy AR, Kowdley KV, Iloeje U, et al. The impact of chronic hepatitis B on quality of life: a multinational study of utilities from infected and uninfected persons. Value Health 2008;11:527-38. [Crossref] [PubMed]
- Pwu RF, Chan KA. Cost-effectiveness analysis of interferon-alpha therapy in the treatment of chronic hepatitis B in Taiwan. J Formos Med Assoc 2002;101:632-41.
- Lam ET, Lam CL, Lai CL, et al. Health-related quality of life of Southern Chinese with chronic hepatitis B infection. Health Qual Life Outcomes 2009;7:52. [Crossref] [PubMed]
- Che YH, You J, Chongsuvivatwong V, et al. Dynamics and liver disease specific aspects of quality of life among patients with chronic liver disease in Yunnan, China. Asian Pac J Cancer Prev 2014;15:4765-71. [Crossref] [PubMed]
- Jia YX, Cui FQ, Li L, et al. Comparison between the EQ-5D-5L and the EQ-5D-3L in patients with hepatitis B. Qual Life Res 2014;23:2355-63. [Crossref] [PubMed]
- Zhuang G, Zhang M, Liu Y, et al. Significant impairment of health-related quality of life in mainland Chinese patients with chronic hepatitis B: a cross-sectional survey with pair-matched healthy controls. Health Qual Life Outcomes 2014;12:101. [Crossref] [PubMed]
- Parmar A, Pal A, Sharma P. National List of Essential Medicines in India: A Story of Deprivation of Substance Use Disorder Treatment. Indian J Psychol Med 2021;43:531-4. [Crossref] [PubMed]
- Ke W, Zhang C, Liu L, et al. Cost-effectiveness analysis of tenofovir disoproxil fumarate for treatment of chronic hepatitis B in China. Hepatol Int 2016;10:924-36. [Crossref] [PubMed]
- Zhang S, Ma Q, Liang S, et al. Annual economic burden of hepatitis B virus-related diseases among hospitalized patients in twelve cities in China. J Viral Hepat 2016;23:202-10. [Crossref] [PubMed]
- Wei L, Li J, Cao Y, et al. Quality of life and care burden in primary caregivers of liver transplantation recipients in China. Medicine (Baltimore) 2018;97:e10993. [Crossref] [PubMed]
- Liu GE. Guidelines for Pharmacoeconomic Evaluations in China 2020. Beijing: China Market Press; 2020.
- Zhang S, Wang C, Liu B, et al. Cost-effectiveness of expanded antiviral treatment for chronic hepatitis B virus infection in China: an economic evaluation. Lancet Reg Health West Pac 2023;35:100738. [Crossref] [PubMed]
- Ren S, Wang W, Lu J, et al. Effect of the change in antiviral therapy indication on identifying significant liver injury among chronic hepatitis B virus infections in the grey zone. Front Immunol 2022;13:1035923. [Crossref] [PubMed]
- Lee H, Kim BK, Jang S, et al. Cost-Effectiveness Analysis of Antiviral Therapy for Untreated Minimally Active Chronic Hepatitis B to Prevent Liver Disease Progression. Clin Transl Gastroenterol 2021;12:e00299. [Crossref] [PubMed]
- Razavi-Shearer D, Estes C, Gamkrelidze I, et al. Cost-effectiveness of treating all hepatitis B-positive individuals in the United States. J Viral Hepat 2023;30:718-26. [Crossref] [PubMed]
- Jeng WJ, Liaw YF. Letter to the Editor: Proper monitoring instead of expanding treatment for improving the prognosis of indeterminate phase hepatitis B patients. Hepatology 2023;78:E93-4.
- Lok AS, Perrillo R, Lalama CM, et al. Low Incidence of Adverse Outcomes in Adults With Chronic Hepatitis B Virus Infection in the Era of Antiviral Therapy. Hepatology 2021;73:2124-40. [Crossref] [PubMed]
- Lee HW, Kim SU, Park JY, et al. Prognosis of Untreated Minimally Active Chronic Hepatitis B Patients in Comparison With Virological Responders by Antivirals. Clin Transl Gastroenterol 2019;10:e00036. [Crossref] [PubMed]
Cite this article as: Zhuo HY, Lin Y, Song HW, Hong MZ, Yao LF, Pan JS. Cost-effectiveness analysis of oral antiviral therapy in patients with indeterminate chronic hepatitis B infection. Transl Gastroenterol Hepatol 2025;10:43.


