
Anatomical S5 segmentectomy for regional cholangitis due to bile duct injury: versatile applications of indocyanine green (with video)
Highlight box
Surgical highlights
• This technique underscores the multifaceted applications of indocyanine green in liver surgery, from preoperative planning and intraoperative guidance to postoperative assessment, thereby enhancing the safety and efficacy of hepatic resections.
What is conventional and what is novel/modified?
• Traditionally, liver resection using blue-dye administration or clamping has been the technique of choice. However, it is often challenging to accurately identify the transection margins.
• The use of indocyanine green (ICG) makes it much easier to clearly delineate the transection line, as well as to identify areas with abnormal biliary drainage, tissue perfusion, or bile leakage.
What is the implication, and what should change now?
• The use of ICG allows for safe and efficient surgery, maximizing healthy liver parenchyma and reducing complications. The application of ICG in liver surgery should be expanded, along with a thorough understanding of its potential complications.
Introduction
Background
Iatrogenic injury to the biliary tract is a very severe complication that can affect both the quality of life and survival of patients (1). Among the many treatments available, the surgical resection of the affected hepatic segment has been described (2). This surgical intervention is very challenging and poses a significant challenge even for expert hepatobiliary and pancreatic surgeons.
Indocyanine green (ICG) is a virtually harmless molecule, approved by the Food and Drug Administration in 1959 (3). Following intravenous administration, it binds to alpha-1 lipoprotein, diffusing through all vascularized tissues in the body (4-6). Subsequently, ICG is taken up into hepatocytes by organic anion transporting polypeptide 1B3 and Na+/taurocholate co-transporting polypeptide transporters and is excreted unchanged into the bile, utilizing the bile salt export pump multidrug resistance-associated protein 2 (7). Upon irradiation with near-infrared light, ICG absorbs the light at 800 nm, emitting a strong fluorescence at a wavelength of 830 nm. This fluorescence can be digitally processed and subsequently displayed on a screen to provide accurate surgical guidance (8,9).
Rationale
In recent years, numerous applications in the field of hepatobiliary and pancreatic surgery have been described for ICG. In 2008, Aoki et al. published the first studies on fluorescence-guided surgery for performing anatomical liver resections, where the characteristics of ICG allowed intraoperative fluorescence-guided cholangiography for biliary tract identification (10). Subsequently, ICG was described as a technique for positive and negative staining in hepatic segmentectomies (11). Positive staining involves the infusion of ICG to stain the region where it has been administered, thus facilitating resection following anatomical principles. Negative staining involves vascular clamping of the hepatic region to be resected and the intraoperative administration of ICG, causing all tissue that should not be resected to illuminate, while the tissue that does not capture ICG is resected.
Furthermore, the preoperative administration of ICG has been used to identify abnormal biliary drainage (12). Since ICG must be eliminated via the biliary route, regions with abnormal drainage retain ICG and can therefore be identified intraoperatively thanks to the accumulation of this molecule in the tissues.
Objective
Based on these distribution and tissue diffusion characteristics of ICG, the objective of this video case study, is to present various applications of ICG to perform an anatomical hepatic segmentectomy. We present this article in accordance with the SUPER reporting checklist (available at https://tgh.amegroups.com/article/view/10.21037/tgh-24-156/rc).
Preoperative preparations and requirements
Following iatrogenic bile duct injury, a thorough preoperative study must be conducted to identify the cause and optimal individualized treatment for each patient. In this clinical case, the following were performed:
- Contrast-enhanced computed tomography (CT) revealed an atrophic region in hepatic segment 5, along with slight dilation of the bile duct adjacent to this segment.
- Magnetic resonance cholangiopancreatography (MRCP) demonstrated metallic clips in the B5s, which drain directly into the common bile duct and were significantly dilated.
- After the preoperative study, an endoscopic approach was attempted via endoscopic retrograde cholangiopancreatography (ERCP) to finalize the diagnosis and attempt to resolve the bile duct obstruction. The endoscopic approach failed to surpass the stenotic point, necessitating a surgical approach through open surgery.
- Preoperative administration of ICG—3 days before the surgical procedure, at a dose of 0.5 mg/kg was administered. This administration serves as a preoperative function test (ICGR15: 3.6%). Additionally, given the abnormal bile duct drainage in the affected hepatic segment, ICG could not be properly excreted from the liver, thus accumulating and theoretically helping identify the affected hepatic segment.
Based on the complementary diagnostic tests, it was decided to proceed with an S5 segmentectomy, not only resecting the damaged portion of segment 5 but also performing a complete segmentectomy to prevent recurrent episodes of cholangitis. In this case, an open approach was chosen. The history of cholecystectomy with BDI, along with multiple episodes of cholangitis, suggested the presence of significant adhesions in the surgical field. Additionally, the need for intraportal ICG injection into the S5 pedicles represented a high-risk maneuver, posing a significant technical challenge if attempted via a minimally invasive approach. Nevertheless, despite its technical complexity, a minimally invasive approach for such resections could be a safe and effective technique under certain circumstances.
The study was conducted in accordance with the Declaration of Helsinki and its subsequent amendments. Informed consent was obtained from the patient. Ethics committee approval was not required, as the procedure was part of routine clinical practice.
Step-by-step description (Video 1)
- Surgical preparation—general anesthesia and orotracheal intubation. Central line cannulation. Positioning with legs closed and arms at the sides. Thompson retractor for cephalic traction.
- L-shaped skin incision—dissection through tissue layers to access the abdominal cavity and perform exploratory laparotomy. Dissection of adhesions in the previous cholecystectomy bed. Upon naked-eye examination, atrophic regions of segment 5 were identified.
- Use of near-infrared imaging with LIGHTVISION NIR Fluorescence Imaging System, SHIMADZU, displayed on an Ikegami screen, showing an atrophic region with altered biliary drainage.
- Delimitation of the area with altered bile duct drainage using monopolar energy.
- Intraoperative ultrasound with ALOKA ProSound Alpha 7, identifying not only bile duct dilations in the atrophic area but also in the non-fluorescing zone surrounding the atrophic segment.
- To minimize the risk of postoperative cholangitis, an anatomic resection of the entire segment 5 was performed using the positive staining technique.
- Guided by intraoperative ultrasound, the medial and cranial branches of the segment 5 portal vein were punctured, followed by the injection of a staining solution consisting of 0.25 mg ICG mixed with 5 mL indigo-carmine. Segment 5 demarcation was slightly visible upon naked-eye examination. Fluorescence imaging clearly delineated the whole segment 5 to be removed with LIGHTVISION, which included the atrophic area identified at the beginning of the surgery.
- Segment 5 was then delimited using monopolar energy (Figure 1).
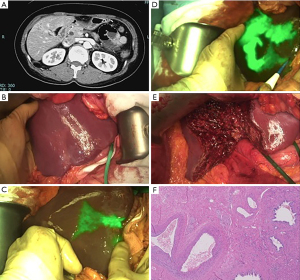 Figure 1 Anatomical S5 segmentectomy. (A) Abdominal CT showing atrophic region in S5. (B) Naked-eye visualization of atrophic region. (C) Near infrared fluorescence imaging showing altered biliary drainage after preoperative ICG administration. (D) Segment 5 demarcation after positive staining with ICG and indigo-carmine. (E) State of the liver after S5 segmentectomy. (F) Anatomopathological findings of sclerosing cholangitis. Hematoxylin & eosin, ×40. CT, computed tomography; ICG, indocyanine green.
Figure 1 Anatomical S5 segmentectomy. (A) Abdominal CT showing atrophic region in S5. (B) Naked-eye visualization of atrophic region. (C) Near infrared fluorescence imaging showing altered biliary drainage after preoperative ICG administration. (D) Segment 5 demarcation after positive staining with ICG and indigo-carmine. (E) State of the liver after S5 segmentectomy. (F) Anatomopathological findings of sclerosing cholangitis. Hematoxylin & eosin, ×40. CT, computed tomography; ICG, indocyanine green. - Under Pringle maneuver, hepatic parenchyma was transected using the clamp-crash technique. During the procedure, the two major portal branches previously stained were identified and ligated. Tributaries of the right hepatic vein and the middle hepatic vein were also divided.
- The root of the dilated hepatic ducts with complete obstruction was identified and divided approximately 1 cm distal to the metal clips placed during the cholecystectomy. The biliary stump was reinforced with a 4/0 non-absorbable suture.
- The specimen was analyzed under near-infrared light, confirming the excision of the entire atrophic area.
- Fluorescence imaging was used to confirm the removal of all stained regions. Subsequently, 1.25 mg of ICG was intravenously injected to confirm blood perfusion of the remaining hepatic parenchyma via fluorescence imaging.
- No bile leaks were identified by naked-eye examination or fluorescence imaging.
- Fibrin glue was sprayed onto the hepatic raw surfaces to ensure hemostasis, and a prophylactic abdominal drain was placed.
- Anti-adhesion material was applied, and the abdomen was closed in a layer-by-layer fashion.
Postoperative considerations and tasks
The postoperative course was uneventful. The patient was discharged on day 8.
The anatomopathological report indicated the presence of sclerosing cholangitis (Figure 1).
Tips and pearls
In this novel technique of fluorescence-guided liver surgery using ICG, various utilities of this molecule have been demonstrated:
- Preoperative function test—ICG was administered preoperatively to assess liver function.
- Identification of atrophic regions—the abnormal bile duct drainage in the affected hepatic segment caused ICG to accumulate, aiding in the identification of the compromised hepatic segment (0.5 mg/kg).
- Intraoperative demarcation. Positive staining—during the surgical procedure, ICG allowed for precise intraoperative demarcation of the affected liver segments through positive staining for segment 5 demarcation (0.25 mg with 5 mL indigo-carmine). The near-infrared fluorescence imaging system enabled clear visualization of the anatomical boundaries of the hepatic segments requiring resection.
- Completion of atrophic area removal—near infrared fluorescence facilitated ensure a complete resection of the altered biliary drainage area was achieved.
- Blood perfusion assessment—post-resection, ICG was used to confirm adequate blood perfusion in the remaining hepatic parenchyma. Intravenous injection of ICG allowed for real-time assessment of liver tissue viability through fluorescence imaging, ensuring that the resected area was limited to non-viable or compromised tissue.
- Detection of bile leaks—ICG fluorescence imaging was instrumental in detecting bile leaks intraoperatively. This immediate assessment allowed for prompt surgical correction, reducing the risk of postoperative complications.
- Enhanced visualization—the integration of near-infrared fluorescence imaging with the surgical workflow provided enhanced visualization of critical anatomical structures, such as bile ducts and blood vessels, improving surgical precision and patient outcomes.
- Another possible approach would have been performing the segmentectomy using negative staining. In this particular case, where there was ICG retention in the damaged part of S5 due to preoperative ICG administration, the use of negative staining could have led to confusion. This is because distinguishing whether the observed fluorescence was due to vascularized tissue not subjected to negative staining or residual ICG retention from the preoperative administration could have been challenging.
This technique underscores the multifaceted applications of ICG in liver surgery, from preoperative planning and intraoperative guidance to postoperative assessment, thereby enhancing the safety and efficacy of hepatic resections.
Discussion
Surgical highlights
In the context of anatomical segment 5 segmentectomy, several critical steps must be meticulously followed to ensure a successful outcome. The preoperative administration of ICG at a dose of 0.5 mg/kg three days before surgery is crucial. This timing allows sufficient uptake and retention of ICG in the liver, particularly highlighting areas with compromised bile excretion (13). Accurate imaging using techniques such as contrast-enhanced CT and MRCP is essential for identifying anatomical abnormalities and planning the extent of the resection (14). During the surgery, the precision of puncturing the medial and cranial branches of the segment 5 portal vein and the subsequent injection of ICG mixed with indigo-carmine for positive staining is paramount. This step ensures the clear delineation of the hepatic segment to be resected (15). The use of the Pringle maneuver to minimize blood loss during parenchymal transection, followed by careful identification and ligation of the major portal branches, is another crucial aspect to consider (16).
Modifications and troubleshooting of the technique
Modifications to the ICG protocol may be necessary based on the patient’s liver function and the specific surgical scenario. For instance, the dosage of ICG might be adjusted in patients with impaired liver function to prevent prolonged retention and potential toxicity (17). The concentration of indigo-carmine can also be modified to enhance the visibility of stained regions. Troubleshooting may involve addressing challenges such as poor visibility of ICG fluorescence, which could be due to suboptimal lighting or technical issues with the imaging system (18). It is essential to ensure optimal conditions for near-infrared imaging, including reducing ambient light and ensuring proper calibration of the imaging equipment. In cases where positive staining does not adequately delineate the target segment, a secondary injection of ICG or adjustments to the imaging parameters may be required (19). Additionally, alternative methods such as using methylene blue dye can be considered if bile leaks are suspected but not detected using ICG fluorescence (20).
Strengths and limitations
Despite its advantages, the use of ICG fluorescence imaging in liver surgery is not without limitations. One primary concern is the variability in ICG uptake and distribution, which can be influenced by the patient’s liver function and metabolic rate (21). In patients with significant liver dysfunction, the visualization might be compromised due to inadequate excretion of ICG. Another limitation is the depth penetration of near-infrared fluorescence, which can affect the visibility of deeper structures within the liver. This limitation may result in the incomplete identification of lesions or abnormalities located deep within the hepatic parenchyma (22). Moreover, there is a risk of false positives or negatives, particularly in detecting bile leaks or identifying compromised tissue. For example, areas with retained ICG may not always correspond accurately with diseased tissue, potentially leading to incomplete resection or unnecessary removal of healthy tissue (23).
Comparison with other surgical techniques and researches
ICG fluorescence imaging represents a significant advancement over traditional methods in hepatobiliary surgery. Unlike standard visual inspection or conventional imaging techniques, ICG provides real-time, intraoperative guidance, allowing for dynamic assessment of tissue perfusion and immediate detection of bile leaks (24). This capability enhances the precision of surgical interventions, reducing the likelihood of postoperative complications. The ability to clearly delineate anatomical boundaries using ICG fluorescence is particularly beneficial compared to relying solely on anatomical landmarks, which can be unreliable due to variations or previous surgical modifications (25). The real-time feedback offered by ICG imaging facilitates more accurate and safer resections, thereby improving overall surgical outcomes (26).
Implications and actions recommended
The potential future applications of ICG fluorescence imaging in liver surgery are extensive. Beyond segmentectomy, this technique could become a standard in various complex hepatobiliary procedures, including tumor resections (27-29), donor liver surgeries, and intricate biliary reconstructions (30). Its utility in visualizing bile ducts and vascular structures could be invaluable in procedures requiring meticulous dissection and reconstruction (31). Additionally, the integration of ICG fluorescence imaging with advanced technologies, such as intraoperative MRI or augmented reality systems, holds promise for even more precise and intuitive surgical guidance. Such integration could further enhance the safety and efficacy of complex surgeries, offering real-time, comprehensive views of the surgical field (32,33).
Conclusions
In conclusion, the utilization of ICG fluorescence imaging in liver surgery offers numerous benefits, including enhanced precision, real-time guidance, and improved patient outcomes. While there are some limitations, such as variability in ICG distribution and limited depth penetration, the technique’s advantages in providing dynamic intraoperative feedback and enhancing surgical accuracy are significant. As technology advances and the understanding of ICG’s applications broadens, this technique is likely to become increasingly integral to a wide range of hepatobiliary surgical procedures. Future developments could expand its use, incorporating advanced imaging modalities and personalized medicine approaches to optimize surgical interventions further.
Acknowledgments
This publication has been made as part of the Doctoral Program in Surgery and Morphologic Sciences of the Universitat Autònoma de Barcelona, Spain.
Footnote
Reporting Checklist: The authors have completed the SUPER reporting checklist. Available at https://tgh.amegroups.com/article/view/10.21037/tgh-24-156/rc
Peer Review File: Available at https://tgh.amegroups.com/article/view/10.21037/tgh-24-156/prf
Funding: None.
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at https://tgh.amegroups.com/article/view/10.21037/tgh-24-156/coif). T.I. reports consulting fees from Kono Seisakusho Co., Ltd. and Tamron Co., Ltd. The other authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. The study was conducted in accordance with the Declaration of Helsinki and its subsequent amendments. Informed consent was obtained from the patient. Ethicsl committee approval was not required, as the procedure was part of routine clinical practice.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Schreuder AM, Busch OR, Besselink MG, et al. Long-Term Impact of Iatrogenic Bile Duct Injury. Dig Surg 2020;37:10-21.
- Lau WY, Lai EC, Lau SH. Management of bile duct injury after laparoscopic cholecystectomy: a review. ANZ J Surg 2010;80:75-81. [Crossref] [PubMed]
- Boni L, David G, Mangano A, et al. Clinical applications of indocyanine green (ICG) enhanced fluorescence in laparoscopic surgery. Surg Endosc 2015;29:2046-55. [Crossref] [PubMed]
- Shafy SZ, Hakim M, Lynch S, et al. Fluorescence Imaging Using Indocyanine Green Dye in the Pediatric Population. J Pediatr Pharmacol Ther 2020;25:309-13. [Crossref] [PubMed]
- Mindt S, Karampinis I, John M, et al. Stability and degradation of indocyanine green in plasma, aqueous solution and whole blood. Photochem Photobiol Sci 2018;17:1189-96. [Crossref] [PubMed]
- Branch RA, James JA, Read AE. The clearance of antipyrine and indocyanine green in normal subjects and in patients with chronic lever disease. Clin Pharmacol Ther 1976;20:81-9. [Crossref] [PubMed]
- Lim C, Vibert E, Azoulay D, et al. Indocyanine green fluorescence imaging in the surgical management of liver cancers: current facts and future implications. J Visc Surg 2014;151:117-24. [Crossref] [PubMed]
- Dai B, Guissi NEI, Sulyok LF, et al. Advantages of using indocyanine green in liver transplantation: a narrative review. Ann Transl Med 2022;10:110. [Crossref] [PubMed]
- Landsman ML, Kwant G, Mook GA, et al. Light-absorbing properties, stability, and spectral stabilization of indocyanine green. J Appl Physiol 1976;40:575-83. [Crossref] [PubMed]
- Aoki T, Yasuda D, Shimizu Y, et al. Image-guided liver mapping using fluorescence navigation system with indocyanine green for anatomical hepatic resection. World J Surg 2008;32:1763-7. [Crossref] [PubMed]
- Ishizawa T, Zuker NB, Kokudo N, et al. Positive and negative staining of hepatic segments by use of fluorescent imaging techniques during laparoscopic hepatectomy. Arch Surg 2012;147:393-4. [Crossref] [PubMed]
- Kawaguchi Y, Ishizawa T, Masuda K, et al. Hepatobiliary surgery guided by a novel fluorescent imaging technique for visualizing hepatic arteries, bile ducts, and liver cancers on color images. J Am Coll Surg 2011;212:e33-9. [Crossref] [PubMed]
- De Gasperi A, Mazza E, Prosperi M. Indocyanine green kinetics to assess liver function: Ready for a clinical dynamic assessment in major liver surgery? World J Hepatol 2016;8:355-67. [Crossref] [PubMed]
- Agostini A, Borgheresi A, Floridi C, et al. The role of imaging in surgical planning for liver resection: what the radiologist need to know. Acta Biomed 2020;91:18-26. [Crossref] [PubMed]
- Chiow AKH, Rho SY, Wee IJY, et al. Robotic ICG guided anatomical liver resection in a multi-centre cohort: an evolution from "positive staining" into "negative staining" method. HPB (Oxford) 2021;23:475-82. [Crossref] [PubMed]
- Mownah OA, Aroori S. The Pringle maneuver in the modern era: A review of techniques for hepatic inflow occlusion in minimally invasive liver resection. Ann Hepatobiliary Pancreat Surg 2023;27:131-40. [Crossref] [PubMed]
- Mai RY, Bai T, Luo XL, et al. Indocyanine Green Retention Test as a Predictor of Postoperative Complications in Patients with Hepatitis B Virus-Related Hepatocellular Carcinoma. Ther Clin Risk Manag 2022;18:761-72. [Crossref] [PubMed]
- Alander JT, Kaartinen I, Laakso A, et al. A review of indocyanine green fluorescent imaging in surgery. Int J Biomed Imaging 2012;2012:940585. [Crossref] [PubMed]
- Jiang Z, Zhou B, Zheng X, et al. A novel method of ultrasound-guided positive staining using indocyanine green fluorescence in laparoscopic anatomical liver resection of segments VII and VIII. Front Oncol 2023;13:1138068. [Crossref] [PubMed]
- Shou-wang C, Shi-zhong Y, Wen-ping L, et al. Sustained methylene blue staining to guide anatomic hepatectomy for hepatocellular carcinoma: Initial experience and technical details. Surgery 2015;158:121-7. [Crossref] [PubMed]
- Nguyen HN, Pertzborn D, Ziadat R, et al. Indocyanine green uptake by human tumor and non‑tumor cell lines and tissue. Biomed Rep 2024;21:136. [Crossref] [PubMed]
- Zhou J, Tan Z, Sun B, et al. Application of indocyanine green fluorescence imaging in hepatobiliary surgery. Int J Surg 2024;110:7948-61. [Crossref] [PubMed]
- Potharazu AV, Gangemi A. Indocyanine green (ICG) fluorescence in robotic hepatobiliary surgery: A systematic review. Int J Med Robot 2023;19:e2485. [Crossref] [PubMed]
- Li XK, Wang P, Su S, et al. Detection of the intraoperative bile leakage in laparoscopic cholecystectomy using indocyanine green fluorescence imaging: A case report. Heliyon 2023;9:e14040. [Crossref] [PubMed]
- Kowalczyk KA, Majewski A. Analysis of surgical errors associated with anatomical variations clinically relevant in general surgery. Review of the literature. Translational Research in Anatomy 2021;23:100107.
- Coco D, Leanza S. Narrative Review on the Use of Indocyanine Green Fluorescence in Surgical Oncology. Maedica (Bucur) 2024;19:807-15. [Crossref] [PubMed]
- Peloso A, Franchi E, Canepa MC, et al. Combined use of intraoperative ultrasound and indocyanine green fluorescence imaging to detect liver metastases from colorectal cancer. HPB (Oxford) 2013;15:928-34. [Crossref] [PubMed]
- Ishizawa T, Fukushima N, Shibahara J, et al. Real-time identification of liver cancers by using indocyanine green fluorescent imaging. Cancer 2009;115:2491-504. [Crossref] [PubMed]
- Huerta M, Gómez-Gavara C, Martínez M, et al. ICG guided surgery enhances liver tumor detection: a complementary tool for intraoperative ultrasonography–a case series study. International Journal of Surgery Open 2024;62:390-4.
- Huerta M, Dalmau M, Fernandes N, et al. Brightening the path to safe liver transplants: the role of ICG fluorescence in biliary anastomosis. BMJ Surg Interv Health Technol 2025;7:e000322. [Crossref] [PubMed]
- Tokuda S, Fujita A, Takagi A, et al. Repair of Bile Duct Injury Using Indocyanine Green Following Laparoscopic Cholecystectomy. Cureus 2023;15:e49312. [Crossref] [PubMed]
- Deng H, Zeng X, Xiang N. Augmented Reality Navigation System and Indocyanine Green Fluorescence Imaging Make Laparoscopic Right Anterior Sectionectomy More Precisely and Safely. J Gastrointest Surg 2023;27:1751-2. [Crossref] [PubMed]
- Chen H, Wu X, Zeng X, et al. The application of augmented reality combined with indocyanine green fluorescence for guiding laparoscopic right hemihepatectomy in the treatment of hepatocellular carcinoma. Eur J Surg Oncol 2025;51:110146. [Crossref] [PubMed]
Cite this article as: Huerta M, Tanaka R, Ishizawa T. Anatomical S5 segmentectomy for regional cholangitis due to bile duct injury: versatile applications of indocyanine green (with video). Transl Gastroenterol Hepatol 2025;10:53.



